
(b) Covalent radii of the elements are shown to scale. The atomic radius for the halogens increases down the group as n increases. However, there are also other patterns in chemical properties on the periodic table. 1: (a) The radius of an atom is defined as one-half the distance between the nuclei in a molecule consisting of two identical atoms joined by a covalent bond. 
Figure: Shell diagram of Mendelevium (Md) atom. Valency electrons : 3: Bohr model: Electron shell for Mendelevium, created by Injosoft AB Md. This similarity occurs because the members of a group have the same number and distribution of electrons in their valence shells. Mendelevium is a chemical element of the periodic table with chemical symbol Md and atomic number 101 with an atomic weight of 258 u and is classed as a actinide. Stability is attained either by losing or gaining or sharing of electrons. The elements in groups (vertical columns) of the periodic table exhibit similar chemical behavior. The main reason for the stability of the noble gas elements is the completely filled orbits of the atom.Įach and every element in the periodic table wants to attain the stability of noble gas as they are the most stable element in the periodic table. As all the orbits of an atom are filled, the new electrons, if added, need a new orbit. These variables allowed Mendeleev to place each element in a certain row (called a period) and column (called a group). These are at the rightmost column of the periodic table. The periodic table is arranged by atomic weight and valence electrons. It draws the dots around the symbol of Palladium. The number of valence electrons an atom has determines its position in the periodic table and its ability to form chemical bonds. The diagram reveals the numbers of Pd valence electrons. We hence have the dot diagram here to simplify the Palladium valence electrons. These are named as the Noble Gas family or the inert gas. Valence electrons are the significant properties of Palladium or any other chemical element. Atoms in Groups 13 and 18 have 3 and 8 valence electrons. For example, atoms in Groups 1 and 2 have 1 and 2 valence electrons, respectively. You can easily determine the number of valence electrons an atom can have by looking at its Group in the periodic table. If every orbit in the atom is completely filled with its maximum number of electrons that it can hold then such atoms are the most stable elements. Valence electrons are the electrons present in the outermost shell of an atom. The group number defines the valence electrons present in the element. Across each row, or period, of the periodic table, the number of valence electrons in groups increases by one from one element to the next. 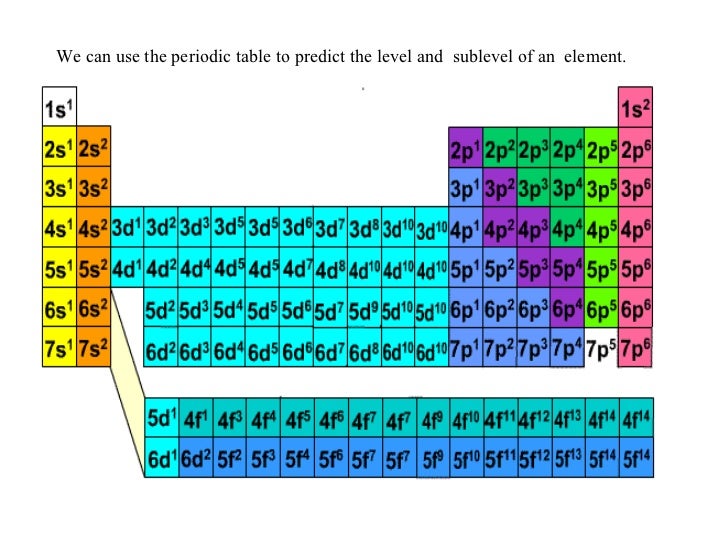
The period number represents the number of shells in an atom. The number of valence electrons in an atom is reflected by its position in the periodic table of the elements (see the periodic table in the Figure below). For elements after No, the electron configurations are tentative.The alkaline metals, the second group of the table, contains two valence electrons, i.e., the outermost orbit of this atom contains two electrons. The electron configurations of the elements indicated in blue are also anomalous, but the reasons for the observed configurations are more complex. The electron configurations of elements indicated in red are exceptions due to the added stability associated with half-filled and filled subshells. \): Electron Configurations of the Elements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
